Key Points:
- Blocking CD38 reduces anxiety-like behavior in middle-aged mice.
- Blocking CD38 improves the learning and memory of middle-aged mice.
- Blocking CD38 increases the wire-hanging capacity of middle-aged mice.
Cognitive decline is associated with mental and emotional deterioration rooted in brain malfunction. Even before the age of 50, measurable decrements in cognitive performance, such as reaction time, impulse control, and learning, can be observed. While the precise cascade of molecular events leading to brain malfunction remains largely unresolved, researchers continue to uncover the influence of NAD+.
CD38 Depletes NAD+
NAD+ is a coenzyme involved in over 500 reactions. It plays an essential role in energy metabolism, DNA repair, and cellular stress resistance. Moreover, there is evidence showing that NAD+ declines with age in multiple organs, including the brain. Thus, considering its vital roles in cellular health and survival, replenishing NAD+ may counteract certain aspects of age-related brain aging and malfunction.
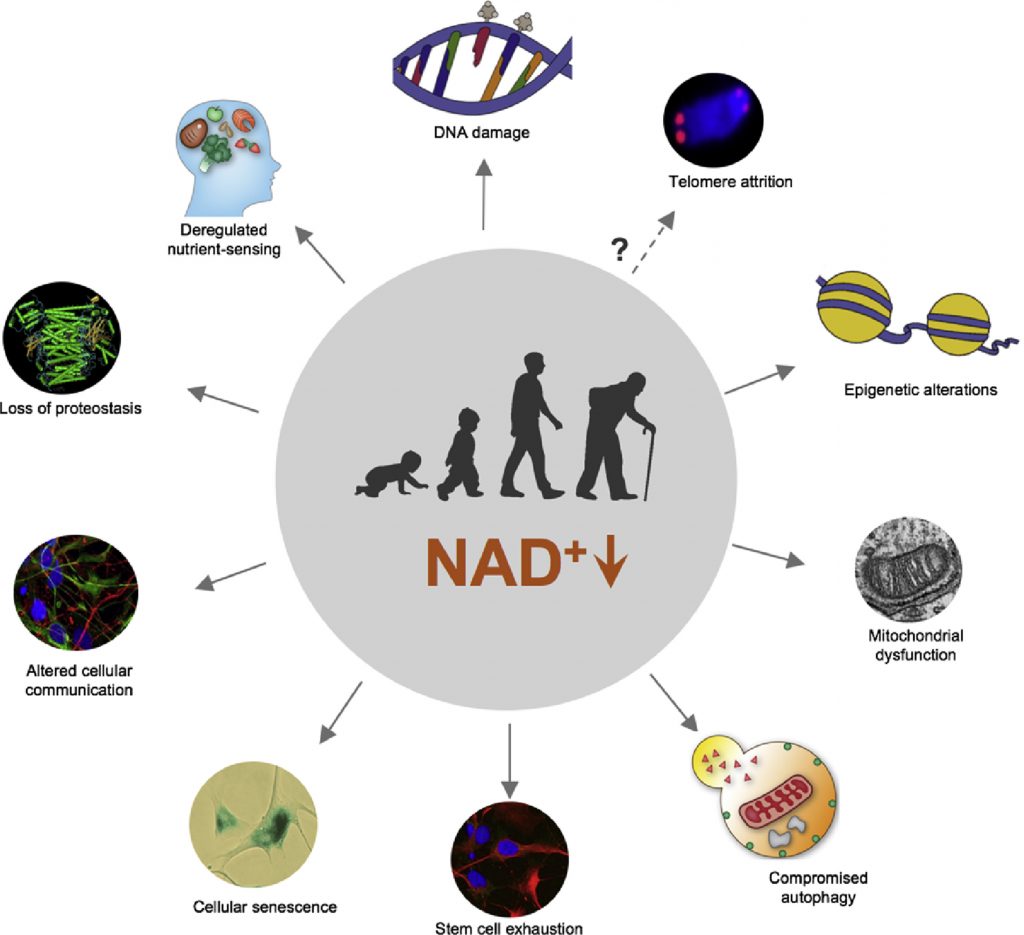
Age-related NAD+ decline is said to be driven by an enzyme called CD38. In mice, blocking CD38 has been shown to prolong lifespan, prevent abnormal heart rhythms, and improve blood glucose levels. Moreover, CD38 is elevated in Alzheimer’s patient brains, and blocking CD38 mitigates symptoms in Alzheimer’s model mice. Furthermore, a gene found in long-lived individuals reduces CD38 activity to elevate NAD+ levels.
CD38 can be found on the surface of immune cells and is known to break down NAD+, leading to lower NAD+ levels. What’s more, when a cell has low NAD+ levels, it can enter a state of cellular senescence. Senescent cells, in turn, secrete pro-inflammatory molecules and activate immune cells equipped with CD38. This generates a vicious cycle of senescence, inflammation, and NAD+ depletion that some say is the primary driver of aging.
An Unlikely Brain Region Associated with Cognitive Decline
While previous studies have shown that blocking CD38 improves cognitive and physical performance in mice, the specific tissues and brain regions involved have not been delineated. The brain is a complex structure surrounded by cerebral spinal fluid (CSF), which cushions the brain from physical impacts, mediates the removal of waste, and nourishes the brain. CSF is produced by a specialized tissue called the choroid plexus.
The choroid plexus usually produces about 16 ounces of CSF per day. However, CSF production declines by up to 50% with aging due to alterations in the choroid plexus. In patients with Alzheimer’s disease and mild cognitive impairment, the choroid plexus is enlarged. Moreover, the larger the choroid plexus is in these individuals, the more severe the decrements in cognitive performance. A larger choroid plexus reflects a swollen, structurally remodeled, inflammatory state. Thus, the choroid plexus seems to play a critical role in brain aging and malfunction.
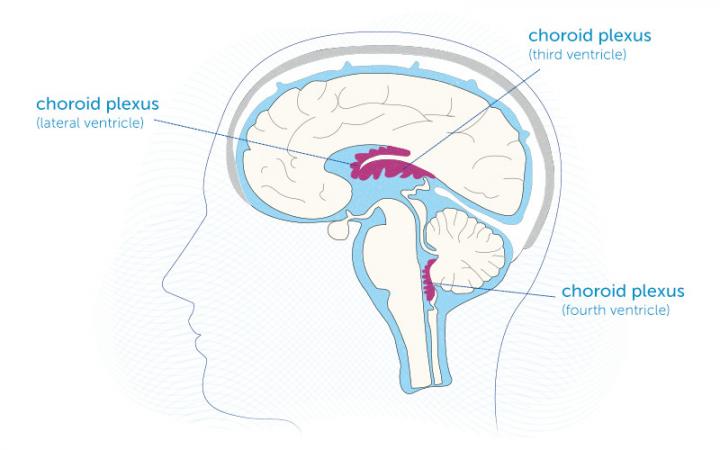
Blocking CD38 Counteracts Choroid Plexus Aging
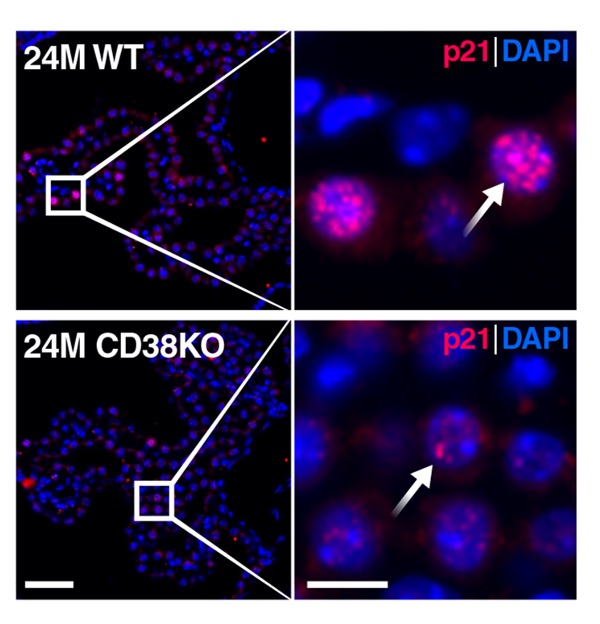
To study the effects of CD38 on aging, scientists from the Buck Institute for Research on Aging in California employed genetically modified mice. These mice, called CD38 knockout mice, were genetically altered to lack the CD38 gene and, by extension, the CD38 enzyme. Through a comprehensive battery of cognitive tests, the Buck Institute researchers showed that, compared to unaltered old mice, old CD38 knockout mice exhibited enhanced learning and memory.
Similar results, where blocking CD38 improves cognition in mice, have been reported previously. However, the specific regions of the brain affected by CD38 were not characterized. Interestingly, the Buck researchers discovered that CD38 levels were much higher in the choroid plexus than in regions of the brain associated with memory, the hippocampus and cortex.
Surprisingly, NAD+ levels were still elevated in the hippocampus of CD38 knockout mice, suggesting that choroid plexus CD38 affects hippocampus NAD+ levels. Additionally, the CD38 knockout mice exhibited less cellular senescence and improved mitochondrial function in the choroid plexus. These findings suggest that blocking CD38 counteracts choroid plexus and hippocampus aging by elevating NAD+, reducing senescence, and promoting mitochondrial health.
Blocking CD38 with New Compound Improves Cognitive and Physical Performance
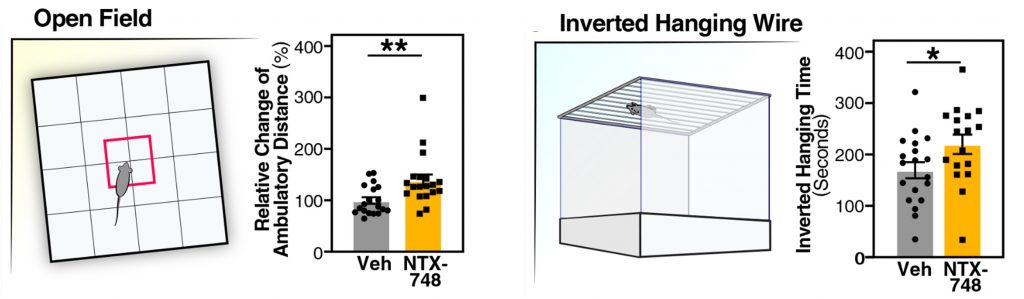
CD38 knockout mice are a valuable experimental tool, but genetically blocking CD38 in human subjects is difficult and likely unnecessary. Furthermore, several molecules are known to block CD38, including apigenin and cyanidin-3-O-glucoside. With this in mind, the Buck Institute researchers developed their own CD38 blocker, called NTX-748. The researchers then supplemented middle-aged mice with NTX-748 for 4 months.
Remarkably, the middle-aged mice supplemented with NTX-748 exhibited less anxiety-like behavior and were able to hang upside-down from a wire longer than normal middle-aged mice, suggesting improved grip strength and muscular endurance. Moreover, similar to the old CD38 knockout mice, the middle-aged mice supplemented with NTX-748 showed improvements in learning and memory. Together, these findings demonstrate that blocking CD38 with NTX-748 can improve cognitive and physical performance.
Furthermore, to assess whether blocking CD38 improves memory at the cellular level, the Buck researchers examined the electrical signals that strengthen connections between neurons. They did so by measuring the electrical activity of the hippocampus, where new memories are formed. The results showed that NTX-748 supplementation increased the strength of this electrical activity, suggesting that NTX-748 strengthens the connections between neurons responsible for memory formation.
Combining Compounds for Synergistic Effects
Synergy occurs when two or more compounds produce a combined effect greater than the sum of their separate effects. The Buck Institute researchers highlight the potential of CD38 inhibition to synergize with other anti-aging interventions, such as:
- Senolytics: Compounds that remove senescent cells, the cells that promote chronic inflammation and organ malfunction. Natural senolytics include fisetin and quercetin, found in plant-based foods.
- NAD+ Precursors: Compounds that elevate NAD+ levels, including nicotinamide, NR (nicotinamide riboside), and NMN (nicotinamide mononucleotide).
Since blocking CD38 prevents the degradation of NAD+ and NAD+ precursors boost NAD+, combining these compounds may synergistically restore NAD+ levels. Moreover, normal NAD+ levels can prevent the generation of senescent cells while senolytics can remove existing senescent cells, suggesting that combining NAD+ restoration with senolytics may synergistically maintain low senescent cell levels.
Future studies exploring the combined effects of these anti-aging interventions will confirm whether blocking CD38, boosting NAD+, and removing senescent cells work synergistically. In the meantime, compounds targeting these aspects of cellular aging can be found separately. However, it may be a while before NTX-748, the CD38 blocker developed by the Buck Institute, is available to the public, and this novel compound may potentially be tested as a new drug for clinical applications.