Key Points:
- Rapamycin reduces the number of times older adults can stand up from and sit down in a chair within 30 seconds.
- While not statistically significant, rapamycin users walked a shorter distance in six minutes than older adults who took a placebo.
- A marker for inflammation, C-reactive protein (CRP), was elevated in rapamycin users, but this was mainly due to two participants having exceptionally high levels.
Rapamycin has long been touted as a promising longevity intervention, although human evidence remains limited. Recently, researchers from New Zealand and the United States examined rapamycin’s effects on exercise performance in older adults. Their study, published in the Journal of Cachexia, Sarcopenia and Muscle, suggests that rapamycin may impair exercise performance.
Rapamycin May Blunt Exercise-Induced Muscle Gains
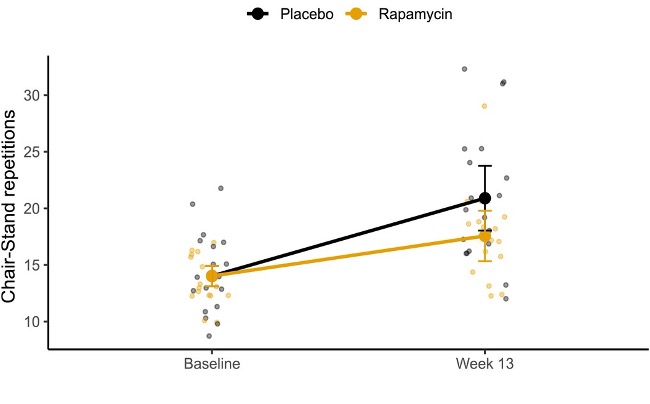
To assess exercise performance, the researchers used the 30-second chair-stand test, in which participants repeatedly stood up from and sat down in a chair for 30 seconds. The test is commonly used as a measure of lower-body strength and fall risk. In this study, sedentary older adults performed chair-stand exercises three nonconsecutive days per week for 13 weeks. Because chair stands are a form of resistance exercise, improvements in strength are reflected in the number of repetitions completed within the same 30-second interval.
During the 13-week exercise regimen, participants received either 6 mg of rapamycin or a placebo once a week. The dose was administered 24 hours after the final exercise session of the week to avoid interfering with the muscle-building response. By the end of the study, both groups improved their performance on the 30-second chair-stand test, but the rapamycin group completed about two fewer repetitions on average than the placebo group. These findings suggest that rapamycin may blunt exercise-induced gains in muscle performance.
Does Rapamycin Worsen Endurance, Strength, and Quality of Life?
The participant performed chair stands as part of a home-based exercise program. The program also included an endurance component, in which participants cycled for up to 25 minutes. At the end of the study, the researchers measured 6-minute walk distance. They found that the rapamycin group walked about 16 fewer feet on average than the placebo group. The rapamycin group also produced about 2.6 pounds less force on a grip strength test, and self-reported health-related quality of life scores worsened. However, none of these negative effects reached statistical significance.
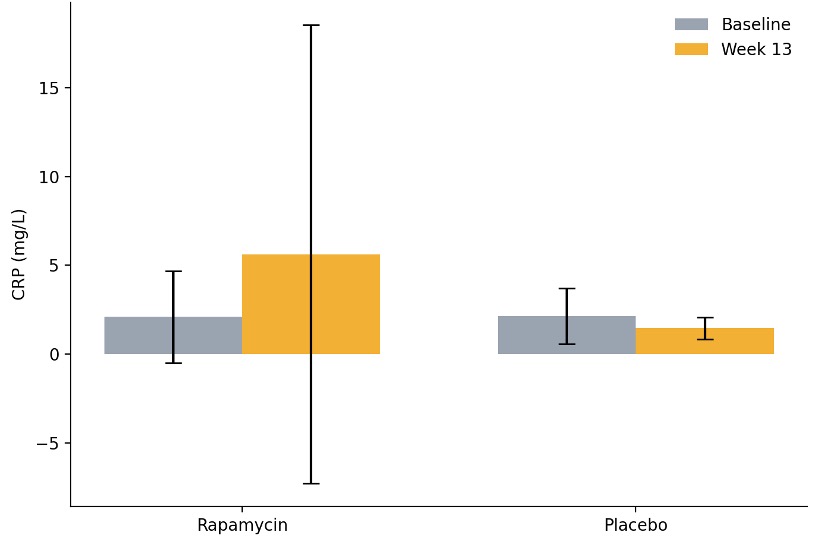
The researchers also assessed inflammation, which is a major driver of aging, and biological age. To measure inflammation, they tested C-reactive protein, or CRP, a marker often used in clinical practice. CRP increased significantly, but the researchers said this was driven by two participants with exceptionally high CRP levels. Additionally, to assess biological age, the researchers used a commercial home kit called TruAge, which estimates biological age from chemical markers on DNA known as epigenetic markers. They found that rapamycin did not affect biological age.
Safety & Adverse Events
The researchers also measured the number of adverse events in both the placebo and rapamycin groups. While there were 99 events reported in the rapamycin group, 63 were reported in the placebo group. The researchers say that the majority of these events were mild, and the most common were headache, fatigue, and upper respiratory tract symptoms.
Ultimately, the adverse events that could be attributed to rapamycin were higher in the rapamycin group than in the placebo group. Notably, one of the participants in the rapamycin group was hospitalized due to acquiring pneumonia. Since rapamycin can suppress the immune system, it could make individuals more susceptible to infections like pneumonia. Overall, these findings suggest that rapamycin may lead to unwanted, potentially life-threatening side effects.
Lab Safety Analysis
Lab tests revealed several differences between the placebo and rapamycin groups. Participants in the rapamycin group had smaller red blood cells and more platelets, which can occur in response to infections or long-term elevated inflammation. They also had higher alkaline phosphatase levels, which indicate potential liver damage, and higher HbA1c levels, which can indicate high blood glucose levels. Moreover, the rapamycin group showed higher LDL cholesterol levels, which is a risk factor for cardiovascular disease. The researchers point out that these results were clinically modest.
Did the Researchers Use Too High a Dose?
Rapamycin was first approved as an immunosuppressant drug for organ transplant rejection. However, longevity biologists have found that rapamycin prolongs the lifespan of rodents more than most other pharmacological interventions. Rapamycin inhibits a nutrient-signaling molecule called mTOR (mechanistic target of rapamycin), which is thought to be hyperactive in old age. The idea is that rapamycin restores mTOR activity to normal levels.
Nevertheless, mTOR is a key modulator of cell proliferation and growth, and is necessary for muscle hypertrophy. A previous study of healthy young men showed that taking 16 mg of rapamycin before resistance exercise reduces the protein synthesis needed to build muscle. To circumnavigate the potential blunting of muscle growth, the New Zealand and United States researchers used a 6 mg/week dose of rapamycin. However, it may be possible that this dose could have been too high.
We have previously reported on studies showing that rapamycin counters heart aging and brain aging in middle- to older-aged adults. Additionally, we reported on an Oxford study showing that rapamycin reduces age-promoting senescent cells in older adults. In these studies, the participants took 1 mg/day of rapamycin, which is the same dose used in a keystone study demonstrating that rapamycin makes older adults less susceptible to flu infection. On the other hand, we reported on a study showing that 10 mg/week of rapamycin improves lean mass in older women. However, in this previous study, the participants took rapamycin for 48 weeks. Ultimately, it seems that the optimal dose and duration of rapamycin treatment needs to be worked out.
Future Large Rapamycin Study
Interestingly, one of the researchers on the study, Dr. Matt Kaeberlein, recently interviewed Dr. Bonnie LaFleur, a scientist working on a large rapamycin study. Dr. LaFleur explains that the study will test the effects of 4 mg/week or 8 mg/week of rapamycin on resilience and immune aging in 720 older adults. This study may help to uncover whether the benefits of supplementing with rapamycin outweigh the side effects.