Key Points:
- Microbiome depletion improves memory and reduces brain inflammation, while reducing age-promoting senescent cells.
- The brain rejuvenating effects of microbiome depletion were recapitulated by a potential new drug.
- Longevity interventions that have been shown to improve gut microbiome health and counteract cellular aging are also listed.
Are the bacteria inhabiting our gut friend or foe? A new pre-print study from Harvard University suggests the latter, at least when it comes to healthy aging. Dr. Lee Rubin and his team showed that depleting the gut microbiome of aged mice counteracts key aspects of unhealthy aging, such as memory loss. However, these findings do not necessarily negate the promise of beneficial bacteria—probiotics—in promoting healthy aging, and we list several longevity interventions that promote a balanced microbiome.
Microbiome Depletion Improves Memory and Reduces Brain Inflammation
To determine how erasing the gut microbiome could affect the brain, Dr. Rubin and his team depleted the gut microbiota of aged mice using a cocktail of antibiotics (ampicillin, neomycin, vancomycin, and metronidazole). Remarkably, this led to improvements in learning and memory, as shown by the novel object recognition (NOR) test. Moreover, the microbiome-depleted aged mice exhibited higher levels of neuronal activation and an increase in the generation of new neurons. These findings suggest that microbiome depletion improves memory by promoting neuroplasticity, the process necessary to form new memories.
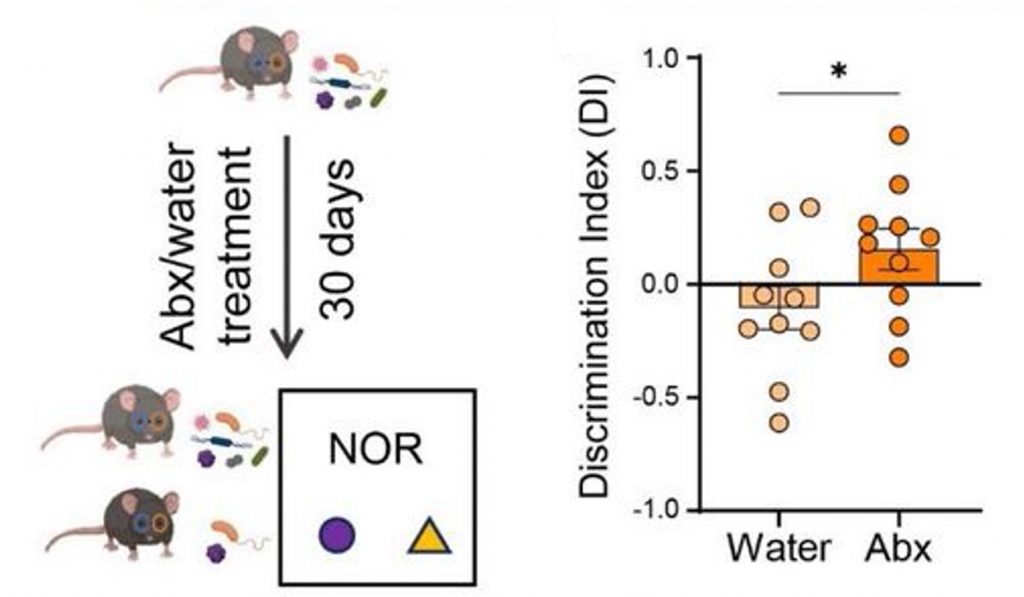
Brain aging, which encompasses mild memory loss, slower processing speed, reduced attention, and decreased brain volume, is caused by the structural deterioration of the brain. For example, the layer of fat that wraps around the axons of neurons, myelin, declines with age, leading to impaired brain connectivity. The blood vessels of the brain also deteriorate, which can allow pathogens to enter the brain and induce inflammation. The researchers showed that microbiome depletion counteracts both blood vessel and myelin deterioration, which could account for the observed improvements in memory and neuroplasticity.
Senescent Cells
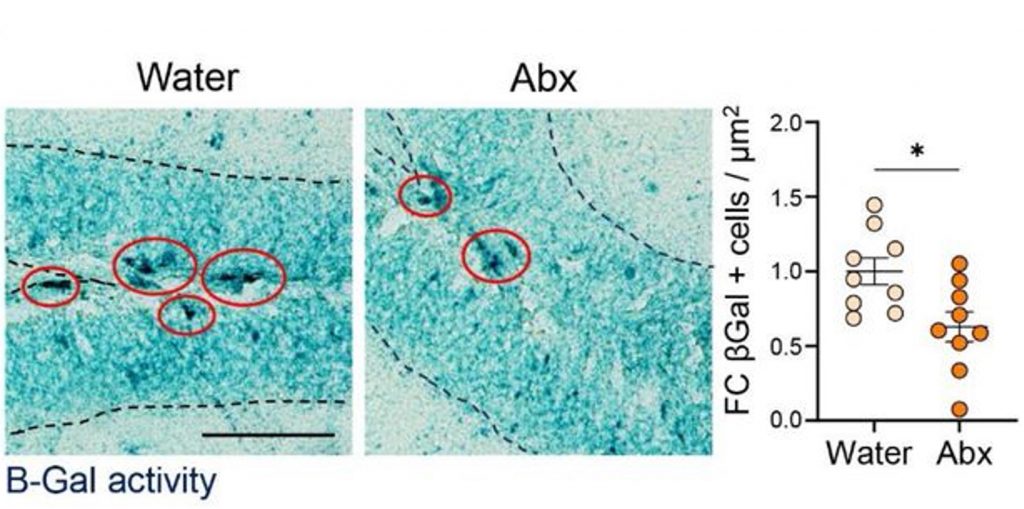
Senescent cells have emerged as one of the key drivers of aging. When our cells experience stress, such as DNA damage, they enter a senescent state. Once senescent, these cells have been found to secrete pro-inflammatory and tissue-degrading molecules that promote tissue and organ aging, including brain aging. It follows that a reduction in senescent brain cells can promote improved memory. Indeed, the researchers found that microbiome depletion led to a reduction in senescent cells within the hippocampus, a region of the brain where memories are formed.
Microglia
Brain inflammation is also generated by microglia, the brain’s resident immune cells. At rest, microglia are mostly harmless, but when they become activated in response to cellular stressors, they begin to secrete pro-inflammatory molecules. Chronically activated microglia contribute to neurodegenerative disorders like Alzheimer’s disease by causing inflammation. Strikingly, the researchers found a reduction in activated microglia in microbiome-depleted aged mice, which was accompanied by a decrease in inflammatory markers. Together, these findings suggest that microbiome depletion decreases brain inflammation by reducing pro-inflammatory cells like senescent cells and activated microglia.
The Retina and Heart
To determine if microbiome depletion affects tissues other than the brain, the researchers examined the retina and heart. These tissues are highly vascularized, meaning they have more access to signaling molecules that travel through the bloodstream from the gut microbiome. Similar to the brain, it was found that the retina had more blood vessels and fewer activated microglia. As for the heart, there was less scarring (fibrosis) and a mitigation of hypertrophy. These findings suggest that microbiome depletion counteracts aspects of aging in multiple tissues, including the brain, retina, and heart.
A Drug that Mimics Microbiome Depletion?
The microbes within our gut secrete signaling molecules that can pass through the gut wall and into the bloodstream. It is these signaling molecules that affect tissues throughout the body, including the brain (if they can cross the blood-brain barrier). To identify these gut-to-brain signals in microbiome-depleted aged mice, the researchers measured changes in circulating molecules that matched changes in the brain. Several factors able to cross the blood-brain barrier were identified, including eotaxin-1, which previous studies have shown is implicated in cognitive aging and neurodegeneration.
The researcher next tested whether inhibiting eotaxin-1 could recapitulate the brain rejuvenating effects reaped from microbiome depletion. To do so, they administered an antibody that blocks eotaxin-1 to normally aged mice. Astonishingly, this led to similar effects as microbiome depletion, including an increase in blood vessels, enhanced formation of new neurons, and reduced activated microglia. Aged mice treated with anti-eotaxin-1 also had more oligodendrocyte stem cells, which are the brain cells that mature into myelin-producing cells. Moreover, blocking eotaxin-1 improved the memory of aged mice, as measured by the NOR test.
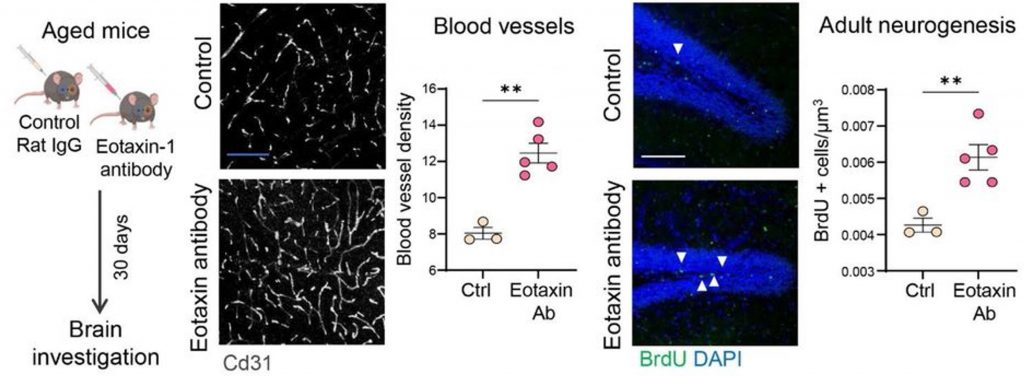
Further experiments suggested that eotaxin-1 is secreted by built-up immune cells called eosinophils in the gut. High counts of eosinophils often indicate an allergic reaction, asthma, or infection. Since depleting the gut microbiome reduces eosinophil numbers, these findings suggest that eosinophils play a large role in brain aging. With that said, depleting the gut microbiome in humans may not be feasible, so eotaxin-1 inhibitors, if developed into a drug, could be a potential alternative.
Longevity Interventions that Restore Gut Microbiome Health
The gut microbiome includes trillions of microbes, including bacteria, viruses, and fungi. Scientists are actively researching which microbes are beneficial and which are harmful. So far, certain strains of bacteria, such as those that digest fiber and secrete a type of fat called short-chain fatty acids (SCFAs), have proven beneficial. However, the findings of Dr. Rubin and his team support the notion that getting rid of harmful microbes can be beneficial as well. It follows that having a balanced microbiome, consisting of minimal harmful microbes and an abundance of beneficial ones (probiotics), may be the key to healthy aging.
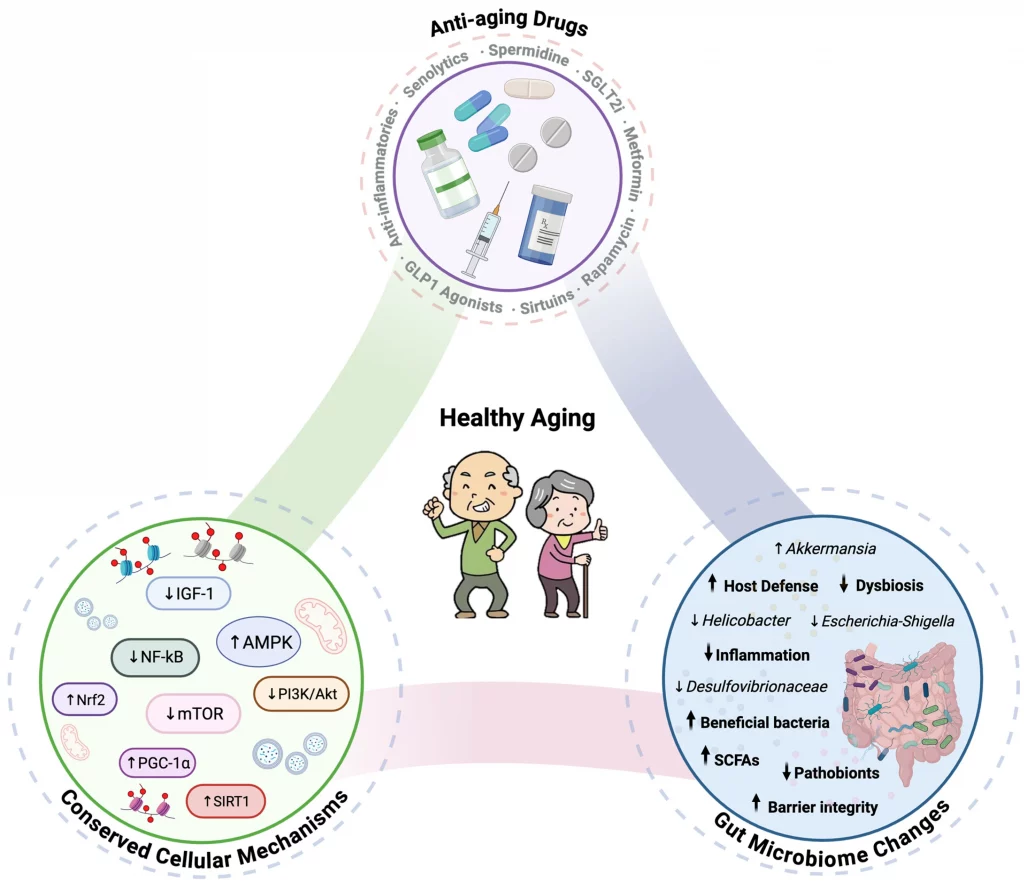
With that being said, there are several longevity interventions that have been shown to improve gut microbiome health. These include senolytics (which get rid of senescent cells), spermidine, SGLT2 inhibitors, metformin, rapamycin, sirtuins, GLP-1 receptor agonists, and anti-inflammatories. These longevity interventions lead to similar improvement in cellular aging, while also counteracting age-related gut microbiome imbalances. Whether the cellular aging improvements triggered by these interventions are the cause or consequence of improved gut microbiome health remains an active area of research.