Key Points:
- Fecal microbe transfer from old to adult mice adversely affects the nervous system, reducing exploratory behaviors and inducing anxiety.
- The transfer of microbes from old to adult mice also reduces immune function.
- This microbial transfer increases biological age and shortens lifespan.
The collective community of microorganisms in the gut (known as the microbiome) communicates with the nervous and immune systems. As organisms age, cellular stress from reactive molecules (oxidative stress) can lead to the deterioration of these systems, the microbiome, and the communication between them. Along these lines, some researchers have suggested that the microbiome itself, with age-related changes in microbial species, plays a critical role in driving the aging process via the breakdown in its communication with, and influence on, the nervous and immune systems. However, unraveling whether age-related changes to microbiome composition are a cause or consequence of aging has remained unclear.
Now, as published in Mechanisms of Ageing and Development, researchers from Complutense University of Madrid in Spain show that transferring gut microbes from old to adult mice accelerates biological aging (an age assessment based on how well cells, tissues, and organs function). Aligning with this result, the researchers also found that this transfer of microbes shortened lifespan. Moreover, this microbial transfer altered the nervous and immune systems to increase anxiety and cause immune decline, respectively. These findings corroborate the notion that age-related changes to the microbiome, in part, drive aspects of aging and suggest that targeting the microbiome may serve as a way to slow aging.
Gut Microbes from Old Mice Adversely Influence the Nervous and Immune Systems
To evaluate whether the transfer of gut microbes from old to adult mice adversely impacts the nervous system, the Spanish researchers performed a behavioral test, since all behavior ultimately stems from the nervous system. They employed the holeboard test, which measures anxiety and exploratory behavior by exploiting the mice’s natural curiosity and propensity to explore new environments. Along these lines, the holeboard test consists of a square arena containing a raised, flat floor with multiple equally spaced holes. The main behavior observed during this test is called head-dipping, where a mouse inserts its head into the hole to explore its surroundings.

In the holeboard test evaluations, the adult mice that received gut microbes from old mice showed significantly reduced head-dipping behavior after 18 months (roughly equivalent to 56 years for humans). Because mice typically have an inclination to engage in exploratory head-dipping behavior, this finding showed that the gut microbes from old mice reduced exploration, likely by increasing anxiety levels. Accordingly, these findings suggest that old gut microbes adversely affect the nervous system.
To assess whether the old gut microbes transferred into adult mice adversely affect the immune system, the Spanish researchers examined immunity-related markers, such as natural killer cell activity. Natural killer cells bind to and destroy cells that are infected with viruses as well as tumor cells. Accordingly, low natural killer cell activity may indicate reduced immunity against infections and cancer.
The researchers found that, after 18 months, adult mice that received gut microbes from old mice exhibited significantly lower natural killer cell activity. This finding suggests that the transfer of microbes from old into adult mice adversely impacts the immune system.
Gut Microbes from Old Mice Shorten Lifespan
To evaluate whether the adverse effects of gut microbes from old mice on the nervous and immune systems of adult mice negatively impact biological age and longevity, the scientists assessed biological age and lifespan. They found that after 18 months, the adult mice that received the old microbe transfers exhibited a 7-week increase in biological age (roughly equivalent to a 16-year increase in humans). Moreover, these mice also showed a significantly shorter lifespan. These findings show that gut microbes from old mice transferred to adults significantly increase biological age and shorten lifespan.
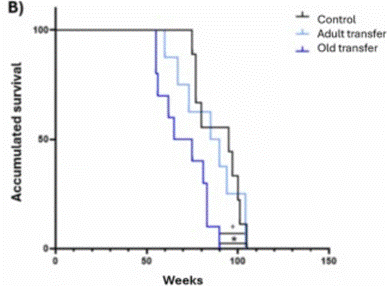
“These findings provide compelling evidence that gut microbiota plays a crucial role in aging and directly influences life expectancy,” say the Spanish researchers in their publication.
Targeting the Microbiome with Supplements or Fecal Microbiota Transplantation
This study provides some of the first evidence that, indeed, age-related changes to the microbiome, to some degree, drive aging, rather than simply arising as a consequence of aging. With these results in mind, targeting the microbiome could serve as a way to slow aspects of aging, especially those that pertain to the nervous and immune systems. For confirmation of this notion, future research needs to confirm that transferring gut microbes from younger into older mice slows aspects of aging and potentially extends lifespan.
To target the microbiome, certain bacterial species, such as Akkermansia muciniphila, accumulate with age in healthy older individuals. Supplements containing this bacterium are available for supplementation, and, based on the findings suggesting that gut microbes influence aging, supplementation with these bacteria could help preserve the nervous and immune systems and slow the aging process.
Another procedure people can use to regain a microbiome composition seen during younger years is fecal microbiota transplantation. This procedure, which is used for certain infections and is under investigation for experimental applications, involves transferring stool from a healthy donor into a recipient’s gastrointestinal tract to restore beneficial gut bacteria. Fecal microbiota transplantation is often performed via colonoscopy, where a thin, flexible tube equipped with a camera is inserted rectally, or by taking pills containing gut microbes. Accordingly, in the future, it may become more common for people to use either of these methods to reconstitute a younger microbiome for the preservation of the nervous and immune systems and to slow aging.