Key Points:
- Oxygen delivery capacity declines with age in humans, contributing to an age-accelerating condition called hypoxia.
- Aging leads to the depletion of inosine in red blood cells to compensate for impaired energy production.
- Inosine supplementation restores oxygen to tissues and improves memory and grip strength in middle-aged mice.
Our survival depends on the survival of our cells, and the survival of our cells depends on oxygen. Now, researchers from Central South University in China have recently discovered that aging disrupts oxygen delivery, according to a new study. They also showed that restoring oxygen delivery with inosine counteracts mental and physical decline. The findings suggest that replenishing inosine can delay aging.
Oxygen Delivery Capacity Declines with Age in Humans
What separates the young from the old, and the old from the very old? A study from last year reveals that oxygen delivery dwindles with age, except for in people who live beyond 90. To confirm this, the Central South University researchers measured oxygen delivery from old and young donors. They examined how strongly the donors’ red blood cells (RBCs) bind to oxygen. The more strongly oxygen binds to RBCs, the less oxygen RBCs can release and deliver to cells. The researchers found that, with increasing age, oxygen binds more strongly to RBCs, which likely reduces oxygen delivery. The findings support the notion that oxygen delivery declines with age.
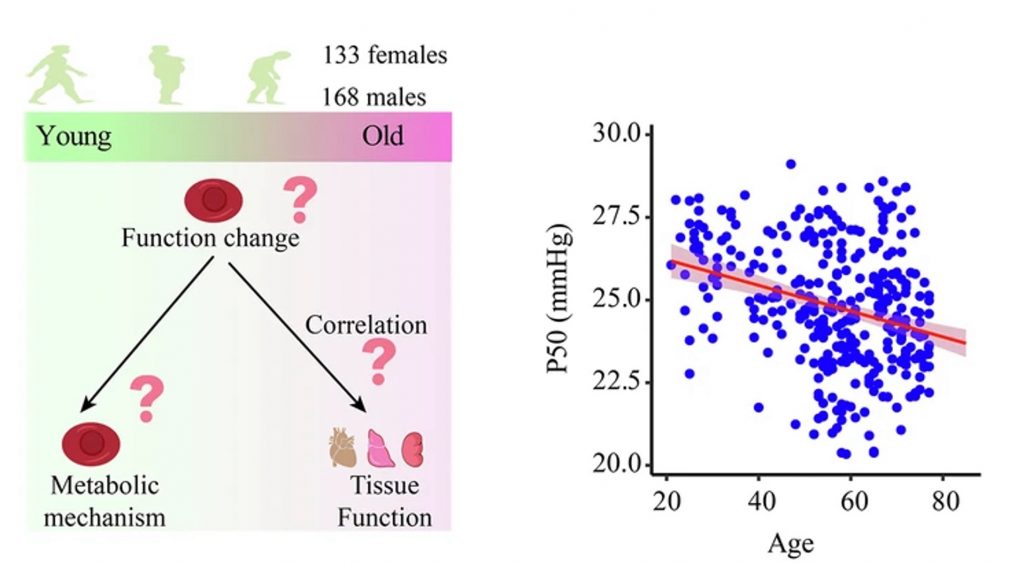
The researchers also showed that declining oxygen delivery correlates with multiple signs of aging. Notably, they demonstrated that P50 decline correlates with cardiovascular, metabolic, liver, and kidney decline. Moreover, at the molecular level, they found that RBCs from older donors exhibited impairments in cellular energy metabolism. They also found that RBCs from older donors are laden with reactive oxygen species (ROS). Studies have shown that ROS contribute to cellular damage and aging. Overall, the researchers surmised that aging alters the way RBCs generate cellular energy, which could trigger elevated ROS levels.
Inosine Supplement Combats Aging in Mice
To investigate further, the researchers explored whether the results found in humans could be found in mice. Consistent with human RBCs, they observed elevated ROS and impaired energy metabolism in mouse RBCs. Through further experimentation with mice and human RBCs, the researchers then elucidated how aging leads to reduced oxygen delivery. Essentially, they found that, due to impaired energy metabolism, aged RBCs utilize inosine as fuel, which depletes inosine. They then came to the conclusion that inosine depletion contributes to reduced oxygen delivery capacity.
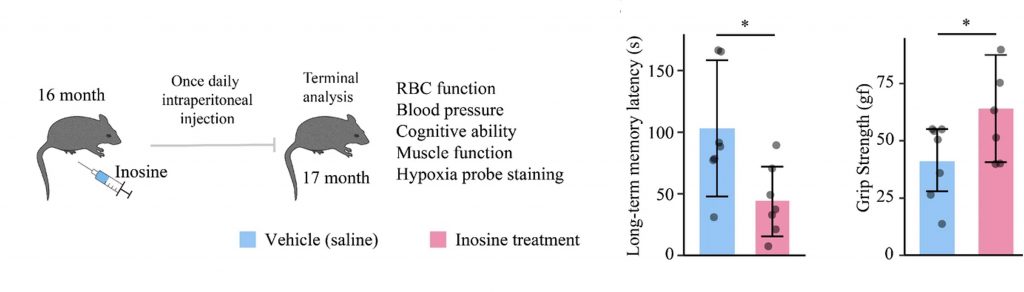
To test if inosine supplementation restores oxygen delivery and counteracts aging, the researchers injected mice with 20 mg/kg/day of inosine. They utilized 16-month-old mice, which model 50-year-old humans. Strikingly, the researchers found that inosine enhanced oxygen delivery capacity, lowered ROS levels, and improved long-term memory and grip strength. However, they found that inosine had no effect on weight or blood pressure. Moreover, in heart, brain, kidney, and muscle tissue, the researchers found reduced oxygen levels, a condition known as hypoxia. Together, these findings suggest that inosine supplementation restores oxygen delivery and counteracts key aspects of aging.
“Overall, our study significantly advances our understanding of aging from a completely new angle, elucidating the dysfunction of RBCs during aging from a metabolic perspective and proposing a novel concept in which tissue damage during aging is a result of the impairment of oxygen release from erythrocytes. By repositioning RBCs as active regulators of healthy aging, we open new avenues for rejuvenation,” said the authors.
Combining Inosine with NAD+ Booster
Studies have shown that NAD+ (nicotinamide adenine dinucleotide) declines with age. Moreover, research demonstrates that NAD+ precursors like NR (nicotinamide riboside) can restore NAD+ levels. Previously, Russian scientists found that an RBC enzyme called PNP (purine nucleoside phosphorylase) blocks the conversion of NR into NAD+. Interestingly, in the present study, the researchers found that PNP is hyperactive in the RBCs of older mice.
This could mean that, in older adults, PNP hyperactivation blocks elevations in NAD+ from NR. However, inosine largely restores RBC metabolism, which could reduce PNP hyperactivity and allow RBCs to convert NR into NAD+. It follows that combining inosine with NR could synergistically raise NAD+ levels in RBCs. Moreover, in many studies, NR counteracts age-related decline, so combining it with inosine may lead to synergistic effects against aging.
One study examined the effect of combining an NAD+ precursor, nicotinamide, with inosine and two other antioxidants, succinic acid and riboflavin. Notably, the study showed that the antioxidant combination alleviated symptoms of nerve damage in type 2 diabetics. Additionally, a study of immune cells showed that NR elevates inosine, and NR and inosine have similar effects on autophagy. Autophagy removes disruptive cellular components, like misfolded proteins, and keeps cells healthy. Altogether, these studies suggest that combining an NAD+ precursor with inosine may synergistically counteract aging at the cellular level.