Key Points:
- Restoring brain NAD+ with a molecule that balances NAD+ metabolism, P7C3-A20, can reverse Alzheimer’s disease (AD)-related memory and learning deficits in mice.
- In symptomatic AD mice, P7C3-A20 can reverse amyloid plaque accumulation, neuroinflammation, and young and mature neuronal cell death.
- NAD+ balance dysregulation correlates with AD severity in mice and humans, and comparing NAD+’s effects on AD mice and humans’ proteins highlights reversal nodes.
For more than a century, people have seen Alzheimer’s as a one-way street—once memory fades and pathology sets in, the brain can only get worse, not better. But a new study shows that may not be the case.
Recent research from Case Western Reserve University suggests late-stage degeneration of the brain (neurodegeneration) may be biologically reversible, at least in certain circumstances. According to research on mice and humans, restoring NAD+ levels may prevent Alzheimer’s and similar neurodegenerative diseases as well as reduce serious symptoms and damage. This work challenges the idea that Alzheimer’s is irreversible and supports treatment development.
NAD+ Down, Neurodegeneration Up
NAD+ is a tiny molecule with a big role in cellular metabolism, aiding cells in producing energy, fixing damaged DNA, controlling inflammation, and managing oxidative stress. Research has shown that as people get older, their levels of NAD+ naturally drop. Previous studies have suggested that these levels drop even more in neurodegenerative diseases like Alzheimer’s. It hasn’t been clear whether that decline is just a side effect or a cause of the disease. Yet, whether restoring NAD+ balance could do more than slow Alzheimer’s damage has been unclear.
To answer those questions, the Case Western researchers looked at NAD+ balance in mice and humans. Specifically, they looked at what happens when NAD+ levels are recovered.
They did not just raise NAD+ levels randomly; they brought them back to a healthy bodily range.
To do so, they used a molecule called P7C3-A20, which stabilizes NAD+ metabolism and has been studied for neuroprotection but never in an Alzheimer’s context.
The team tested P7C3-A20 on two of the most popular mouse models of Alzheimer’s disease: 5xFAD and PS19. These two models differ slightly in terms of the toxic protein buildup that triggers the Alzheimer’s-like symptoms. When left untreated, both mice’s brain NAD+ levels fell by 30% in the moderate stages and nearly 45% in the advanced stages. That drop was linked to a huge range of Alzheimer’s-related issues from the level of cells to brain function (cognition), notably oxidative damage, disrupted brain cell connections (synapses) in an area crucial for learning and memory (hippocampus), inflammation, and worsening memory.
Balanced NAD+, better aging brains
The researchers discovered that mice that had already begun to exhibit Alzheimer’s symptoms (6 months) had reversed cognitive deficits after receiving P7C3-A20 for six months. These mice demonstrated improved performance on memory tests like object and cue recognition. These mice had stronger hippocampus synapses, indicating cell-level learning. The researchers found that P7C3-A20 reduced the buildup of toxic proteins in both mouse models as well as oxidative stress, neuroinflammation, and DNA damage in Alzheimer’s disease.
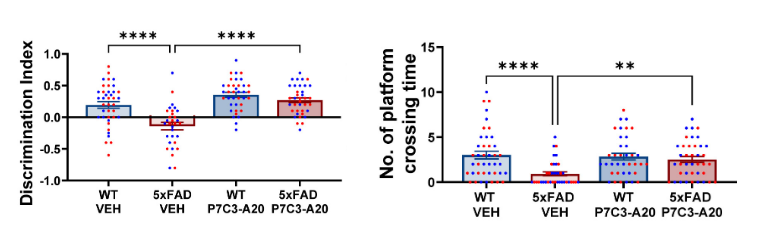
Also, the blood–brain barrier (BBB), which protects the brain from microbes and toxins in the blood, usually breaks down early in Alzheimer’s and lets harmful molecules into the brain, but it also showed signs of structural and functional recovery. Levels of p-tau217, a plasma biomarker that is becoming more common in the diagnosis of Alzheimer’s disease in people, were also lower.
The most interesting finding is that in 11-month-old mice, which were far deeper into their Alzheimer’s development, treatment for just a few weeks improved cognition and brought several molecular markers back to normal, even though mutant tau was still present.
Put together, the researchers said that the study indicates that early Alzheimer’s symptoms may result from diminished brain resilience rather than widespread neuron loss, as 5xFAD mice developed memory and behavioral impairments before significant cell death occurred. NAD+ appears central to this resilience: when levels are disrupted, the brain struggles to cope with toxic protein buildup, but when restored, it supports energy metabolism, proper folding of proteins, mitochondrial function, and inflammation control. This resilience-based approach contrasts with most Alzheimer’s drug strategies, which aim to clear toxic proteins to trigger downstream recovery. Notably, P7C3-A20 did not reduce toxic protein levels; instead, it strengthened the brain’s ability to repair damage and maintain function under stress.
How about in humans?
The researchers then looked into whether these results in mice carried over to humans. To do so, they looked at human brains after death. Alzheimer’s patients had low NAD+ balance, which was linked to toxic protein buildup, oxidative damage, inflammation, synaptic loss, and neuronal deterioration. In contrast, people with amyloid plaques but no cognitive issues had indicators of a healthy NAD+ balance.
Using large protein databases, researchers identified 46 proteins altered in both mouse and human Alzheimer’s brains that returned to normal when treated mice recovered, many linked to mitochondrial function, RNA metabolism, protein quality control, lipid handling, and immune signaling—processes long associated with the disease and now unified by metabolic resilience.
Still, important caveats remain. First, mouse models will never fully capture late-onset Alzheimer’s in humans. Second, only human brain samples from end-stage disease were analyzed. Third, the compound P7C3-A20 has not yet been clinically tested. Also, the researchers did not report the same types of data for both of the Alzheimer’s mouse models. For example, two different methods were used to demonstrate disruption in NAD+ balance between the two models, a choice that was not made clear.
Even so, the findings challenge the idea that Alzheimer’s inevitably progresses through irreversible neuron loss, suggesting instead that failing cellular maintenance systems may be restored. If confirmed in people, this approach could shift treatment from merely slowing decline to rebuilding metabolic resilience and potentially restoring function.