Key Points:
- Early stage pancreatic cancer cells contain senescent cells with high levels of Cox2 — an enzyme that promotes inflammation.
- Treatment with the senolytic drug ABT-737 eliminates these Cox2 senescent cells.
- Mice treated with ABT-737 exhibit significantly less precancerous growth in the pancreas, suggesting that eliminating Cox2 senescent cells halts the progression of cancer.
Cellular senescence remains one of the key drivers of aging and contributes to the progression of multiple deadly age-related diseases. Thus, scientists have focused heavily on a class of drugs called senolytics, known to selectively target and eliminate these harmful cells. One of the most powerful senolytics to date is the compound ABT-737, and new evidence highlights its potential in mitigating pancreatic cancer.
Published in the journal Pancreas, researchers from Israel examined the effects of ABT-737 on the progression of pancreatic cancer in a mouse model with rapid pancreatic cancer growth. Kolodkin-Gal and colleagues showed that senescent cells within precancerous lesions facilitate tumor growth, in part, through Cox2 activation. Additionally, the investigators demonstrated that ABT-737 successfully eliminates these Cox2-activated senescent cells. Notably, clearing out senescent cells with periodic ABT-737 treatment led to drastically less lesion growth. Taken together, the study’s findings suggest that ABT-737 hampers the development of pancreatic cancer and could potentially be used to halt the progression of other senescent-driven cancers.
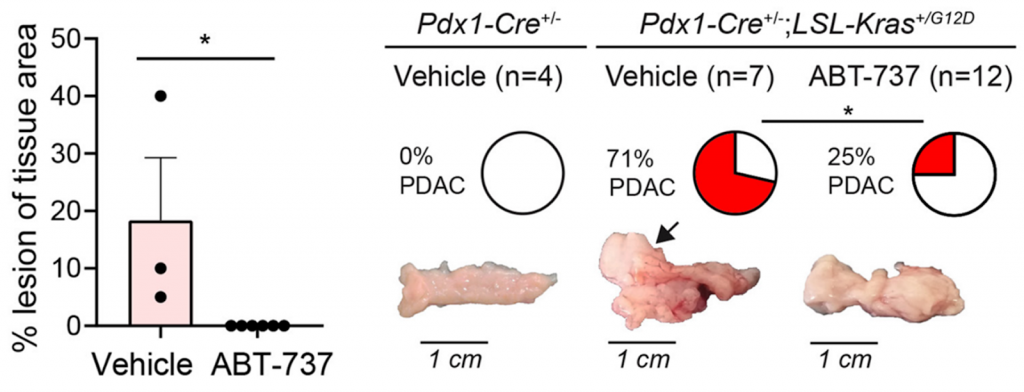
ABT-737 Hampers Lesion Growth
Kolodkin-Gal and colleagues examined whether ABT-737 treatment could hamper precancerous pancreatic lesion growth by treating a mouse model for pancreatic cancer with 25 mg/kg ABT-737. They showed that ABT-737-treated mice exhibited significantly less lesion growth compared to untreated mice, indicating that ABT-737 hinders precancerous lesion development. Next, the researchers analyzed the early stages of tumor generation and whether ABT-737 could affect the development of precancerous lesions into cancer. They found that only 25% of ABT-737-treated mice developed cancer, while 71% of the untreated mice displayed large tumors and increased pancreas size. Taken together, the findings highlight ABT-737’s ability to mitigate the development of pancreatic cancer.
Blocking Cox2 Activity Prevents Lesion Growth
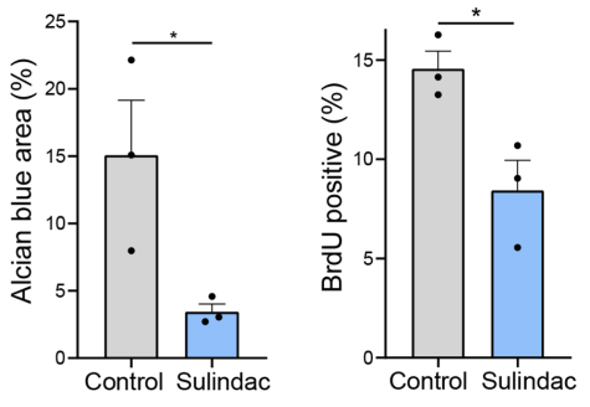
Senescent cells normally serve as a preventative measure to cancer development by hindering the proliferation of cancerous cells. Be that as it may, senescent cells secrete harmful molecules collectively known as the senescence associated secretory phenotype (SASP), which are known to contribute to tumor development (tumorigenesis). By eliminating senescent cells, SASP factors that contribute to tumor growth can also be eliminated.
To determine which SASP factors could be contributing to lesion growth, Kolodkin-Gal and colleagues examined precancerous pancreas cells from the mice modeling rapid lesion growth. They found high levels of the SASP factor Cox2, known to promote inflammation and disease progression. They next sought to determine if Cox2 activity could affect lesion growth. To test this, the investigators supplemented the mice with a Cox2 inhibitor called sulindac via drinking water. Results showed that sulindac treatment halted the proliferation of precancerous cells and led to a 5 fold decrease in lesion size. These results demonstrate that Cox2 activity does indeed stimulate precancerous lesion growth.
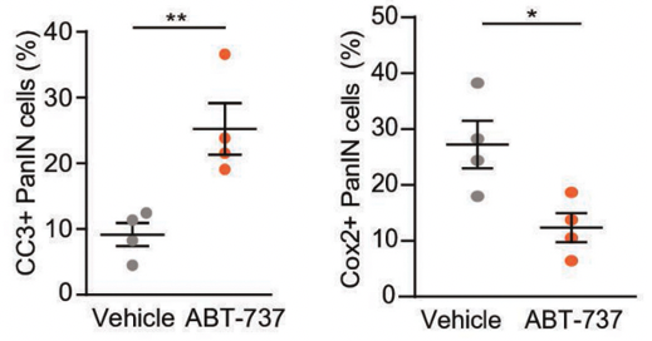
ABT-737 Eliminates Senescent Cells
ABT-737 has been previously studied in other cancer models and is known to inhibit the Bcl2 family, a group of anti-cell death proteins that contribute to senescent cell survival. The investigators found that Bcl2 proteins reside in the same precancerous lesions as Cox2 senescent cells, indicating that ABT-737 could potentially eliminate the senescent cells responsible for lesion growth by targeting the Bcl2 pathway. Following treatment with ABT-737, mice exhibited increased activity of the cell death protein CC3, confirming that inhibiting the bcl2 pathway with ABT-737 mediates senescent cell death in precancerous lesions. Moreover, ABT-737-treated mice displayed significantly less precancerous pancreatic cells with Cox-2 activity compared to control mice, indicating that ABT-737 eradicates Cox2 senescent cells through Bcl2 inhibition.
Are Senolytics The Solution to Cancer and Age-Related Diseases?
Harnessing the power of senolytics continues to shed light on the versatile nature of these drugs, with studies already confirming their effectiveness in boosting muscle regeneration, alleviating cognitive dysfunction and even ameliorating depression in rodent models. Now, this study along with a few others are demonstrating the ability of senolytics to slow down cancer development. Additionally, given that senescent cells are tied to multiple age-related diseases, senolytics have the potential to simultaneously treat various age-related ailments. While these findings are promising, senolytic research in humans is still in its infancy and warrants further investigation to confirm the safety and efficacy of these compounds.