Key Points:
- The senolytic ABT-263 enhances the ability of mesenchymal stem cells (MSCs) to generate cartilage, the deterioration of which leads to arthritis.
- ABT-263 treatment eliminates senescent cells – non-proliferating cells linked to aging – from MSCs by inducing cell death (apoptosis).
- MSCs treated with ABT-263 grow into larger colonies, indicating a higher potential to grow cartilage.
Knee osteoarthritis (OA) is an age-related degenerative joint disease characterized by the progressive loss of cartilage, essential connective tissue that supports many body parts, including our bones, joints, ears, and noses. Recently, scientists have focused on harnessing the power of bone joint MSCs to attenuate the age-related wear and tear of cartilage that leads to OA. In fact, studies have already confirmed MSCs can hamper the progression of OA in rats. That being said, MSCs used for treatment often contain harmful senescent cells that negatively affect the function of surrounding cells, ultimately limiting their therapeutic potential. Thus, limiting senescent cell burden in MSCs is critical for the optimization of MSC therapy against OA.
In a new article published in the journal Stem Cell Research & Therapy, Kentaro Endo and colleagues boost the function of MSCs taken from five OA patients using senolytics – compounds that selectively target and eliminate senescent cells. Following treatment with the powerful senolytic ABT-263, MSCs exhibited improved cartilage and fat cell growth, both of which are vital in maintaining healthy knee tissue. What’s more, MSCs treated with ABT-263 had better colony formation abilities over untreated MSCs and displayed fewer senescent cells than untreated MSCs.
ABT-263 Promotes Joint Cartilage and Fat Growth
What makes MSCs extremely valuable is their ability to become multiple cell types (bone, fat, cartilage). With this in mind, Kentaro Endo and colleagues sought to determine whether the senolytic ABT-263 could improve cartilage and fat cell growth by stimulating MSCs taken from OA patients. To evaluate ABT-263’s effect on cartilage growth (chondrogenesis), the investigators cultured MSCs in the presence of chemicals that stimulate their transformation into cartilage cells (chondrocytes). To measure cartilage growth, the investigators looked at the size and weight of the cartilage pellets that had formed in response to chemical stimulation. Results showed that MSCs treated with ABT-263 had larger cartilage pellets with a mean weight of 4.8 mg, which was 2.9 mg more than untreated MSCs.
Next, investigators examined ABT-263’s effects on fat cell growth (adipogenesis) by growing MSCs with fat cell (adipocyte) promoting chemicals. Three weeks after colony formation, the Tokyo researchers found that treating MSCs with ABT-263 drastically increased the percentage of fat cell growth. Taken together, the findings suggest that ABT-263 could improve joint health by restoring cartilage and fat tissue.
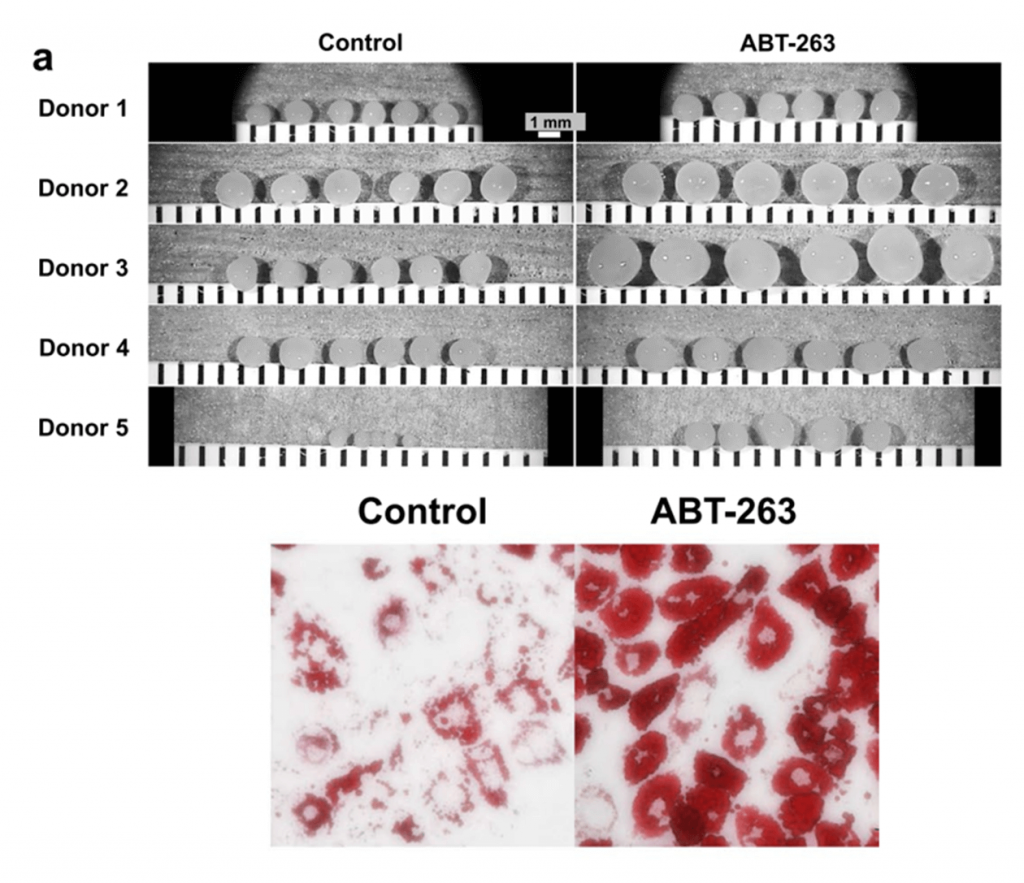
ABT-263 Elevates MSC Proliferation by Limiting Senescent Cell Burden
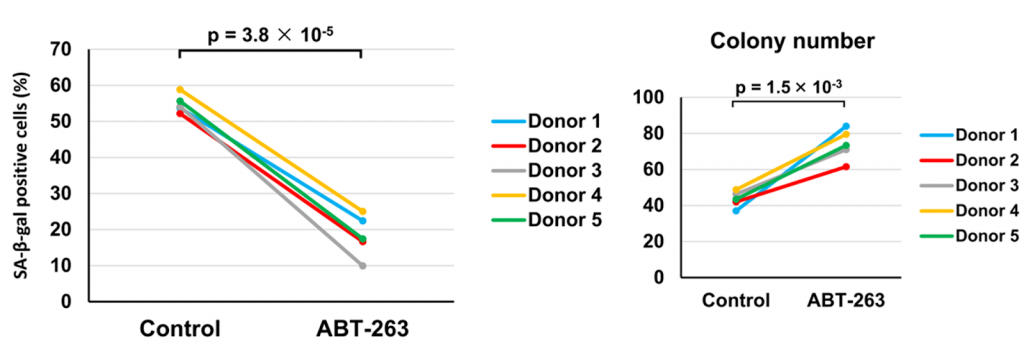
Although ABT-263 has well established senolytic capabilities, investigators examined ABT-263’s effect on senescent cells in OA MSCs. They found that the average percentage of senescent cells in ABT-263-treated MSCs was 18.2% while it was 54.8% in untreated MSCs, demonstrating ABT-263’s senolytic potency. Notably, MSCs treated with ABT-263 had increased activity of an apoptotic protein called cleaved caspase-3, indicating that ABT-263 eradicates senescent cells through induction of apoptosis.
Healthy stem cells proliferate (divide and multiply) before becoming other cell types, promoting the growth of tissue like fat and cartilage. Following ABT-263 treatment the investigators saw that MSCs displayed significantly larger colonies than untreated MSCs, indicating elevated proliferative capacity. Furthermore, ABT-263 treated MSC colonies contained fewer senescent cells, suggesting that senescent cells inhibit MSC proliferation. Overall, these findings suggest that ABT-263 treatment leaves more healthy cells to transform into vital cartilage and fats cells needed to ameliorate OA deficits.
The Power of Senolytics
Senolytics have a multifaceted nature that has sparked some excitement in the field of aging. Given that senescent cell accumulation occurs across the body and contributes to the progression of various age-related diseases, senolytics have the potential to target multiple aging-linked pathways simultaneously. Additionally, senolytics hold anti-aging properties, with studies confirming that they can alleviate inflammation and cognitive dysfunction in rodents. Although the science has barely scratched the surface of human applications for senolytics, the available data suggest that they hold great potential as safe and effective aging therapeutics.