Key Points:
- Mice subjected to the most severe form of sepsis — septic shock — exhibit 85% higher survival rates when treated with NAD+.
- NAD+ reduces a pro-inflammatory protein called IL-6, associated with promoting aging.
- Treatment with NAD+ reduces the infiltration of immune cells within multiple organs.
While our immune system guards us from bacterial infection, it progressively falters with age. By the time we grow old, our immune system becomes more likely to evoke a life-threatening inflammatory response — sepsis — when threatened by bacterial invaders. However, researchers from Harvard and the Huntington Medical Research Institute may have found how to suppress this uncharacteristic immune response.
As reported in a pre-print (not yet peer-reviewed) from BioRXiv, Iske and colleagues show that NAD+ prevents septic shock-induced death. They also show that NAD+ reduces inflammation while reducing the infiltration of immune cells in multiple organs. These findings suggest that NAD+ blunts the overactive immune response that characterizes sepsis.
NAD+ Can Prevent Death from Inflammatory Shock
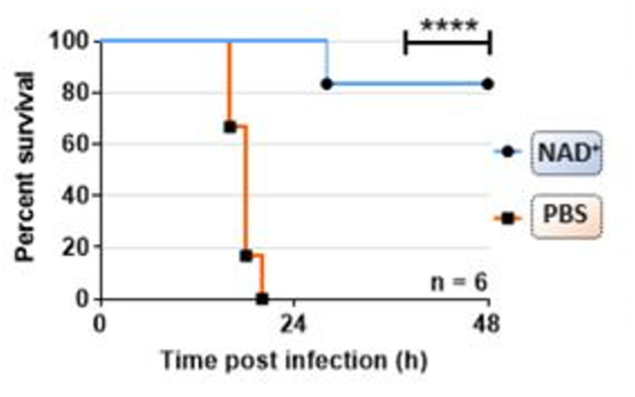
Lipopolysaccharide (LPS) is a molecule found in bacteria like E. coli known to induce septic shock. To test the effect of NAD+ on septic shock, Iske and colleagues treated mice with 40 mg of NAD+ for 2 days followed by a lethal dose of LPS. As result, 85% of the mice survived, whereas all the mice not treated with NAD+ died within 24 hours of infection. These data suggest that NAD+ can prevent death from septic shock.
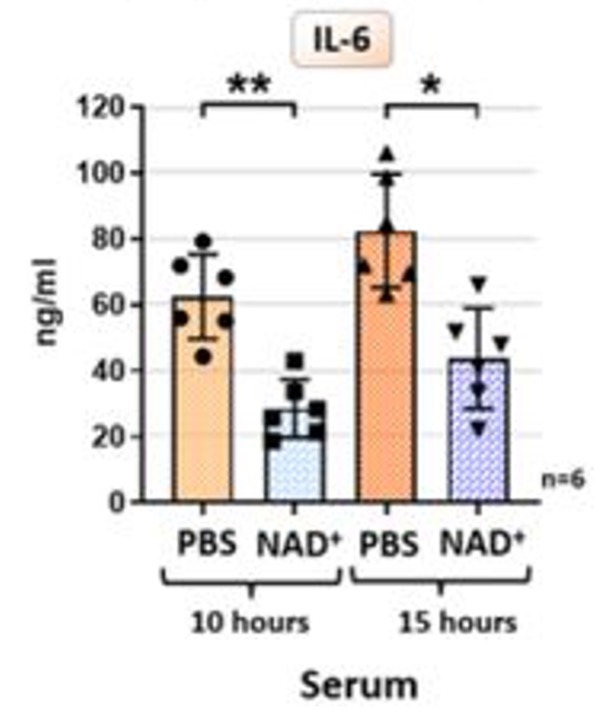
To find if the protective effects of NAD+ arise from suppressing inflammation, Iske and colleagues measured IL-6. It was found that elevations in IL-6 levels induced by septic shock were nearly abolished by NAD+ treatment. These findings suggest that NAD+ could increase survival from infection and septic shock by suppressing overactive immune responses.
Our first line of defense against infection comes in the form of neutrophils — a type of white blood cell. Neutrophils travel to and burrow into infected organs — infiltration — to resolve infection. However, with aging, neutrophils can remain within organs and promote organ destruction via inflammation. Iske and colleagues found that NAD+ reduced neutrophil infiltration in the lung, liver, and kidney of septically-shocked mice.

While the reduced neutrophil infiltration shown is likely due to fewer neutrophils entering these organs, as opposed to leaving these organs, it is possible that NAD+ could also promote neutrophil migration out of our organs during aging. However, additional studies are needed to confirm this.
Can NAD+ Counter Aging by Regulating Inflammation?
The findings of Iske and colleagues suggest that NAD+ can increase survival from septic shock by suppressing inflammation. Another study has shown similar results using NAD+ nanoparticles. Furthermore, the NAD+ precursor nicotinamide mononucleotide (NMN) has been shown to reduce brain inflammation and improve memory following septic shock.
From these studies, it seems that boosting NAD+ levels modulates the immune system in a way that prevents inflammation from destroying the body. Since chronic inflammation from a dysregulated immune system is known to contribute to aging — inflammaging — it is possible that one of the ways NAD+ counters aging is by reducing inflammation.