Key Points:
- The Nampt activator P7C3 lowers blood glucose while increasing insulin-secreting pancreatic β cells in diabetic mice.
- Treatment with P7C3 increases physical endurance, as well as muscle strength and size.
- P7C3 raises “good” cholesterol and lowers “bad” cholesterol.
As the human population rises and the elderly population expands, unprecedented numbers of individuals with age-related diseases will need treatment. One of these age-related diseases is type 2 diabetes, which can cause other diseases like cardiovascular disease and neurodegenerative disorders. Type 2 diabetes also contributes to sarcopenia — the age-related decline in muscle mass and strength. It is perhaps no wonder why scientists are working hard to find treatments for this pervasive metabolic disease.
Researchers from the University of South Florida report in the Journal of Cachexia, Sarcopenia, and Muscle that they can improve symptoms of diabetes and muscle decline with a molecule called P7C3. They show that P7C3 lowers blood glucose and increases β cells — the cells of the pancreas that secrete insulin — in a mouse model for diabetes. Furthermore, P7C3 increases muscle strength and size while improving physical endurance in these diabetic mice. Moreover, fat metabolism was enhanced and cholesterol levels improved by P7C3.
Mending The Pancreas and Skeletal Muscle
Insulin is a signaling molecule that tells our cells to intake glucose from the bloodstream. In type 2 diabetes, the secretion of insulin by pancreatic β cells is impaired, preventing the normal utilization of glucose and leading to high blood glucose. Manickham and colleagues showed that treating diabetic mice with P7C3 increases insulin levels and the proportion of β cells in the pancreas. Furthermore, P7C3 reduces blood glucose levels, suggesting that P7C3 can reverse the major harms of diabetes.
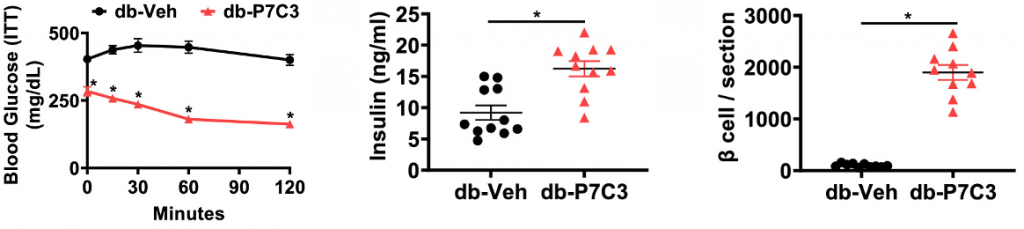
When our cells cannot properly utilize glucose, they cannot use it for energy, and without energy, our cells become dysfunctional and eventually die. This is just one way diabetes can exacerbate age-related diseases like muscle decline. Skeletal muscle is a major consumer of glucose, utilizing it for muscle contraction. Manickham and colleagues showed that diabetic mice exhibit muscle weakness, small muscle cells, and reduced voluntary running, all of which were reversed by P7C3, suggesting that PC73 can reverse diabetes-related muscle decline.
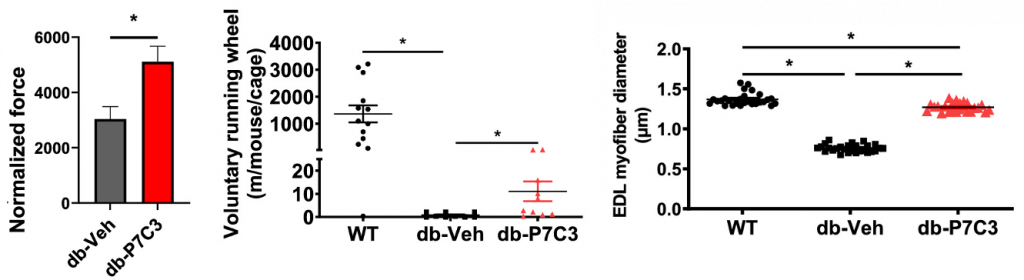
Low HDL and high LDL cholesterol are associated with type 2 diabetes and an increased risk of cardiovascular disease. Manickham and colleagues showed that P7C3 treatment increases HDL and decreases LDL cholesterol in diabetic mice, which could reduce the chances of heart disease. It was also shown that P7C3 improved fat metabolism, leading to less muscle fat, which is associated with poor-quality aged muscle and muscle decline. Overall, these findings show that activating Nampt to boost nicotinamide adenine dinucleotide (NAD+) could result in the reversal of diabetes and muscle decline.
Mending the Brain
Nampt is the key enzyme responsible for synthesizing NAD+, a molecule essential for the production of cellular energy (ATP). NAD+ levels decline with age in many rodent tissues, and in the blood and skin of humans. This decline is associated with many age-related diseases, including diabetes, muscle decline, and neurodegeneration. Thus, restoring NAD+ levels could mitigate these age-related diseases. While there are several ways to boost NAD+, activating Nampt is one of them. P7C3 was initially discovered as scientists searched for ways to enhance the formation of new neurons and was subsequently found to improve the cognitive performance of aged rats.
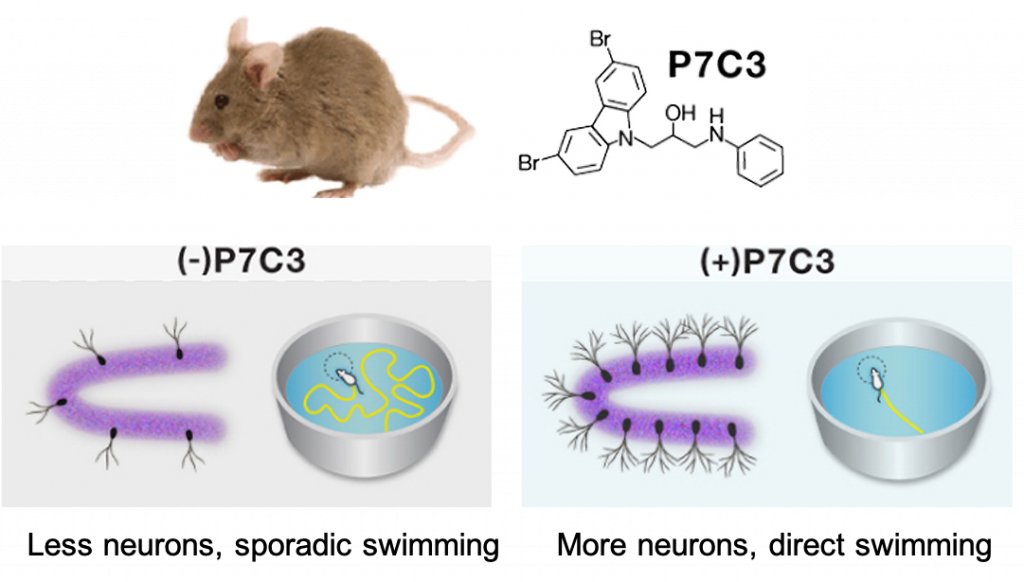
Can Boosting NAD+ Treat Diabetes in Humans?
While the NAD+ booster nicotinamide failed to treat type 1 diabetes, possibly causing insulin resistance due to oxidative stress, other NAD+ precursors — molecules that can increase NAD+ levels — may be more promising. A recent study showed that vitamin B3 lowered blood glucose and reduced the neurodegeneration associated with blindness in diabetic rodents. Nicotinamide riboside (NR) was shown to reverse diabetes-induced heart dysfunction in mice. However, NR lowered Nampt levels and did not increase skeletal muscle NAD+ levels in men with obesity and insulin resistance. Still, nicotinamide adenine dinucleotide (NMN) has been shown to treat diabetic mice. Overall, an apparent lack of human studies with NAD+ precursors like vitamin B3, NR, and NMN to treat diabetes calls for more evidence.