Key Points:
- Researchers screened numerous compounds made by soil bacteria for the ability to affect the build-up of amyloid beta (Aβ) – a toxic protein that is a hallmark of Alzheimer’s.
- The screen identified Verrucarin A, which reduced Aβ production by 80% in a cell-based Alzheimer’s model.
- These results suggest that the interactions between soil organisms and Aβ may provide valuable insight into Alzheimer’s pathology and potentially lead to new therapies.
Modern medicine has exploited the medicinal properties of microorganisms since Alexander Fleming discovered penicillin from the fungus Penicillium notatum in 1928. However, discoveries like these were less frequent until the turn of the century, when advancements in biotechnology and chemistry led to a revival in natural therapeutics. Now, the potential benefits of microorganisms and their metabolites have returned to the forefront of scientific discovery.
A recent study out of Japan, published in Scientific Reports, showed that compounds produced by bacteria in soil could affect amyloid beta (Aβ) production in cells from Alzheimer’s patients. These findings could have significant ramifications for Alzheimer’s and other neurodegenerative diseases, which currently have no cures and are notoriously difficult to treat and prevent.
Screening for Metabolites in the Dirt
Kyoto University and RIKEN researchers screened 98 secondary metabolites isolated from 65 bacteria and 33 fungi found in soil. They utilized neurons reprogrammed from Alzheimer’s disease patient skin cells (induced pluripotent stem cells (iPSC)-derived cortical neurons). These programmed brain cells are used to study sporadic Alzheimer’s disease, accounting for nearly 90% of those with Alzheimer’s disease. The cause of sporadic Alzheimer’s is largely unknown, as these patients do not have a known family history of the disease.
The researchers treated the neurons with metabolites from the soil and measured the production of amyloid beta. Often the amyloid beta aggregates seen in Alzheimer’s consist of amyloid beta 42, even though the amyloid beta 40 concentration is several fold higher. The ratio between the two in the cerebrospinal fluid – the fluid that cushions and protects the brain and spinal cord – has been shown to help diagnose Alzheimer’s patients. By measuring the concentrations of these two forms of beta amyloid in the treated neurons, the researchers identified nine compounds that hit their screening criteria threshold.
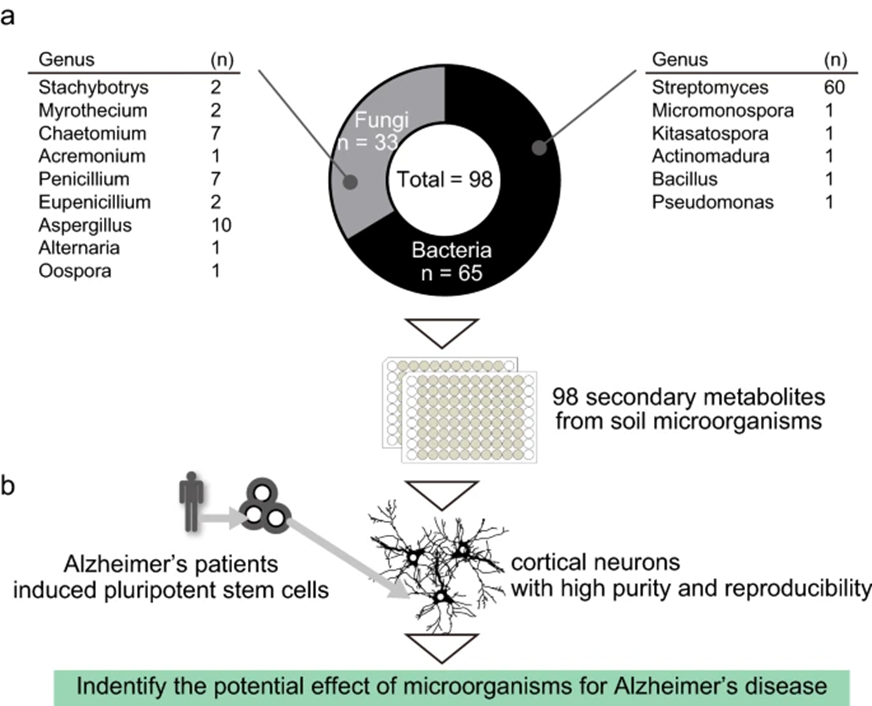
(Kondo, T. et al., 2022 | Scientific Reports) Identifying the Effects of Microorganism Metabolites on Amyloid Beta Production. Screening 98 metabolites from different bacteria and fungi using neurons obtained through inducing pluripotency in stem cells from Alzheimer’s patients and then re-differentiating them into neurons to identify those metabolites that may positively affect amyloid beta production with minimal cellular toxicity.
Screen Identifies Amyloid Beta Reducing Compounds From Bacteria and Fungi in Soil
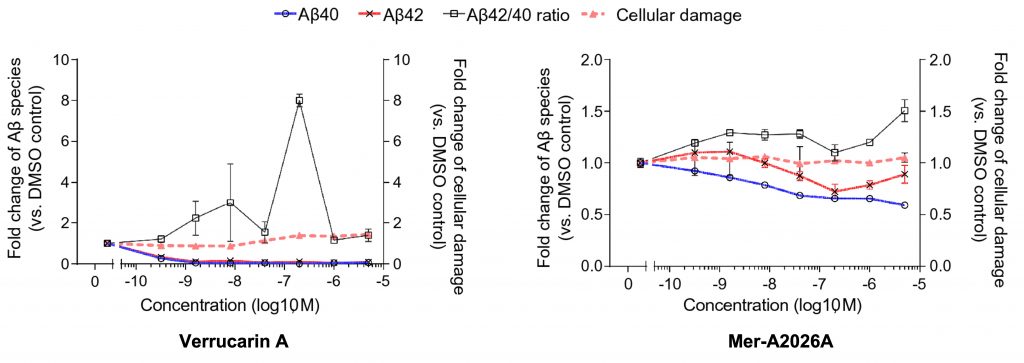
Of the nine compounds that met the screening criteria, the investigators ultimately found two that affected amyloid beta in a dose-dependent manner – meaning that amyloid beta decreased with greater doses. One of the compounds, Verrucarin A from Myrothecium spp., is a known antibiotic that inhibits protein synthesis. Verrucarin A reduced both types of amyloid beta production even at low concentrations.
When used at the appropriate dosage, another compound, Mer-A2026A – a known blood vessel dilator (vasodilator) – was also shown to reduce both forms of amyloid beta with limited cellular damage. Previous research has indicated that reducing amyloid beta could protect against Alzheimer’s onset or progression. Thus these findings demonstrate the potential of Verrucarin A and Mer-A2026A as anti-amyloid therapeutic agents.
Familial Alzheimer’s Neurons Respond Similarly
Occasionally, sporadic and familial Alzheimer’s neurons react differently to the same compounds. For this reason, Kondo and colleagues used another Alzheimer’s model based on familial Alzheimer’s, the inherited form of Alzheimer’s disease. Verrucarin A was shown to suppress amyloid beta production in familial Alzheimer’s neurons, showing its effectiveness regardless of the type of Alzheimer’s. Mer-A2026A also reduced the production of amyloid beta in the familial Alzheimer’s neurons. These findings demonstrate that both Verrucarin A and Mer-A2026A may have therapeutic effects in both sporadic and familial Alzheimer’s disease.
Can Environmental Microorganisms Be the Next Source of Therapeutic Interventions for Alzheimer’s?
Soil microorganisms are only found in certain places at specific times of the year. Exposure to specific microorganisms can even change based on an individual’s age and place of residence. This study suggests that the microorganisms found in our environment could affect the production of proteins in our brain and even affect how our brain ages by potentially reducing Alzheimer’s neurodegeneration, an age-related disease.
While it may seem as if reducing beta amyloid is always beneficial, researchers have found that amyloid beta may play a defensive role in our immunity. Amyloid beta deposition results from infection and inflammation, where the formed aggregates can trap bacteria. And if that’s the case, these compounds may be reducing amyloid beta production to break through our bodies’ defenses. This could increase our susceptibility to infection. Overall, environmental microorganisms and their effects seem to be two-sided. Researchers must still determine whether the benefits outweigh the risks. Is the benefit of decreased amyloid beta (and thus reduced Alzheimer’s risk) worth the risk of potentially increasing our susceptibility to other bacteria – the non-beneficial sort? These microorganisms may only be beneficial to a certain subset of Alzheimer’s patients, but that would still be a major addition to the tool kit for fighting neurodegenerative diseases.