Key Points:
- A gene called NDI1 is delivered to mouse and human cell models for age-related macular degeneration (AMD) as therapy.
- NDI1 therapy reduces molecules called reactive oxygen species (ROS) while raising nicotinamide adenine dinucleotide (NAD+) levels.
- Treatment with NDI1 recovers the function of mitochondria — key modulators of cell health and aging.
To visualize the fine details in life, a layer of tissue coating the back of our eye called the retina must remain intact. However, with aging and a disease called macular degeneration, a region of the retina called the macula slowly degenerates. This age-related macular degeneration (AMD) causes blurred vision that can eventually lead to blindness, yet treatments are lacking.
Now, researchers from Trinity College in Ireland present data in Clinical and Translational Medicine that suggest gene therapy could be used to treat AMD. Millington-Ward and colleagues demonstrate that delivering a gene called NDI1, essential for mitochondrial function, to mice modeling AMD reduces ROS while raising NAD+ levels, reversing the effects of aging. Furthermore, in a human cell model for AMD, NDI1 gene therapy recovers the function of mitochondria.
“Critically, this study provides the first evidence in models that directly modulating bioenergetics in eye cells can provide benefit and improve visual function in dry AMD,” says the senior author, Dr. Farrar.
Gene Therapy Repairs Damaged Retina Cells
As we age, our mitochondria become dysfunctional and begin to produce excessive ROS, which causes damage to cells. Mitochondrial dysfunction and ROS contribute to the retinal degeneration that characterizes AMD. Therefore, Millington-Ward and colleagues experimented with raising the levels of a gene called NDI1, which produces an enzyme needed for mitochondria to function, in a mouse model for AMD. Results showed that NDI1 gene therapy reduced ROS and increased relative NAD+ levels in the retina of these mice, suggesting a reversal of age-related dysfunction.
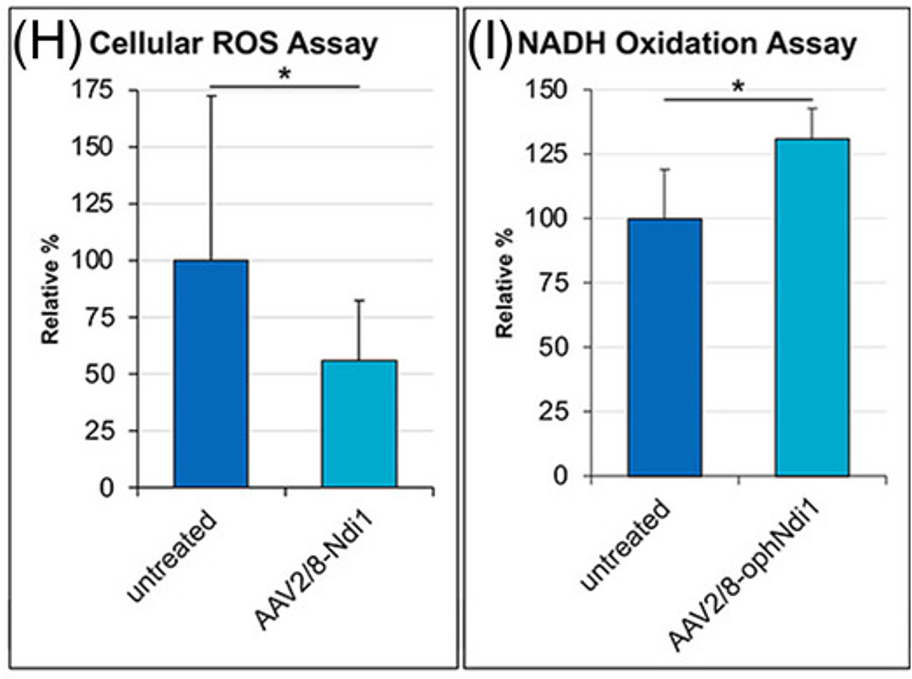
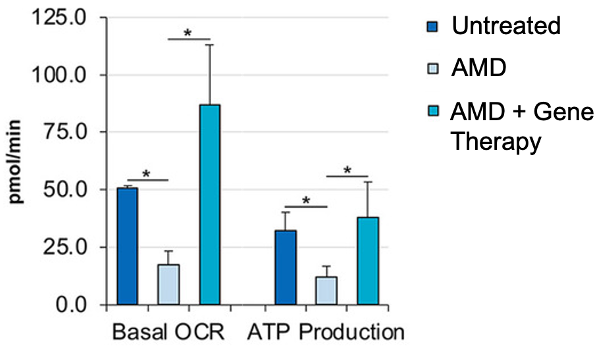
The primary function of mitochondria is to utilize the oxygen we breathe to produce cellular energy called ATP. When mitochondria become dysfunctional, they consume less oxygen and produce less life-sustaining ATP. To establish a human cell model for AMD, Millington-Ward and colleagues exposed human retina cells to a chemical that causes catastrophic damage to the retina. The resulting AMD model cells display deficits in mitochondrial function, including reduced oxygen consumption and ATP production. By treating the human AMD retina cells with the ND1I gene, mitochondrial function was fully restored.
How Does Gene Therapy Work?
In general, gene therapies involve introducing a gene to cells to improve cellular function, often in the hopes of treating a disease. Genes can be delivered using the machinery of a type of virus called adeno-associated virus (AAV), whereby the harmful viral bits are removed and replaced with a beneficial gene. Once the gene makes it into the cell via AAV delivery, the cell uses its own machinery to process and activate the gene.
Using AAV technology, Millington-Ward and colleagues delivered the NDI1 gene to damaged retina cells. The NDI1 gene provides the genetic blueprint for an enzyme (complex I) used by mitochondria to produce ATP. This enzyme also generates NAD+, a vital molecule that mediates the conversion of the food we eat into ATP. Thus, delivering NDI1 to damaged retinas revives mitochondria, allowing them to produce more ATP.
“Many retinal cells, essential for vision, require particularly high levels of energy compared to most other cells, which makes them particularly vulnerable to mitochondrial dysfunction. The therapy we are developing directly targets mitochondrial function and increases energy production levels in the retina, which leads to better visual function in disease models of dry AMD,” said the first author Dr. Millington-Ward.
Is Gene Augmentation a Future Anti-Aging Therapy?
There are currently twenty-four gene therapy products approved by the FDA, including a gene therapy drug called Luxturna used for the treatment of inherited retinal disease. While there is no gene therapy approved for AMD, there are currently two clinical trials (NCT03144999 and NCT03846193) underway for the treatment of dry AMD (not to be confused with wet AMD). The two therapies being tested do not target mitochondria, like NDI1.
Interestingly, NDI1 therapy may not be specific to AMD as mitochondrial dysfunction, excessive ROS, and reduced NAD+ levels underly many age-related diseases, especially neurodegenerative disorders. Mitochondrial dysfunction and excessive ROS are considered hallmarks of aging and, along with low NAD+ levels, underly tissue degeneration in general. While NDI1 still needs further testing, it’s possible that it and similar genes could contribute to treating multiple age-related diseases if used as a therapeutic.