Key Points:
- The lifespan of age-accelerated mice is extended by approximately 15% with cellular reprogramming.
- Cellular reprogrammed mice have less fat, more lean mass, increased strength, and younger skin.
- Markers of arthritis, osteoporosis, and other age-related diseases are marked down by cellular reprogramming.
While breaking the spell of aging is a ceaseless endeavor, scientists are getting closer to making age reversal a reality with cellular reprogramming. Cellular reprogramming has captured the imaginations of many for its remarkable age-reversing capabilities, including reversing the biological age of human cells by about 30 years. Now, scientists are perfecting the craft by honing in on how to reverse the age of mice.
As reported in Aging Cell, Alle and colleagues from the University of Montpellier in France report that cellular reprogramming increases the lifespan and improves the fitness of age-accelerated (progeria) mice. In addition, cellular reprogramming rejuvenates multiple organs, including the metabolic system, skin, bone, lung, spleen, and kidney.
Cellular Rejuvenation Prolongs Lifespan & Reverses Aging
Alle and colleagues induced whole-body cellular reprogramming in a mouse model for progeria, a disease that causes premature aging. Compared to non-reprogrammed mice, the survival rate of reprogrammed mice was longer higher, suggesting that cellular reprogramming increases lifespan.
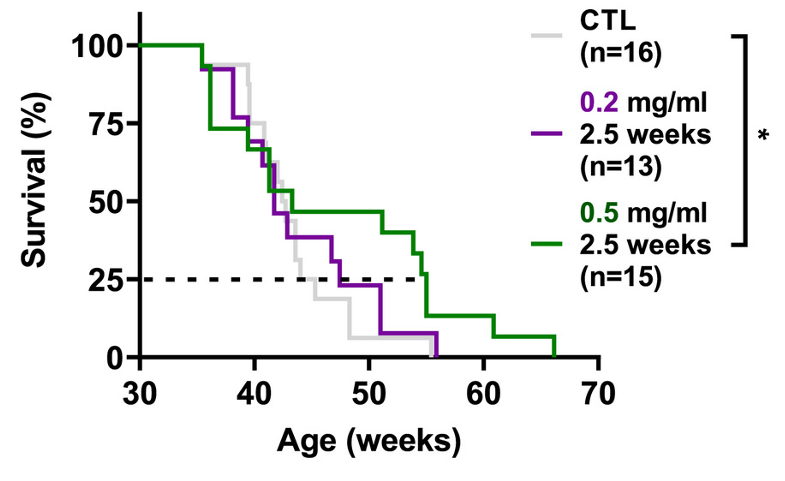
Mice can be genetically engineered to harbor genes containing an “on-switch.” When these mice drink an antibiotic called doxycycline (DOX), the switch is flipped on to activate DOX-controlled genes. Alle and colleagues used the DOX system to turn on Yamanaka factors (Oct3/4, Sox2, Klf4, c-Myc), a set of four genes that trick cells into regressing to an earlier developmental stage.
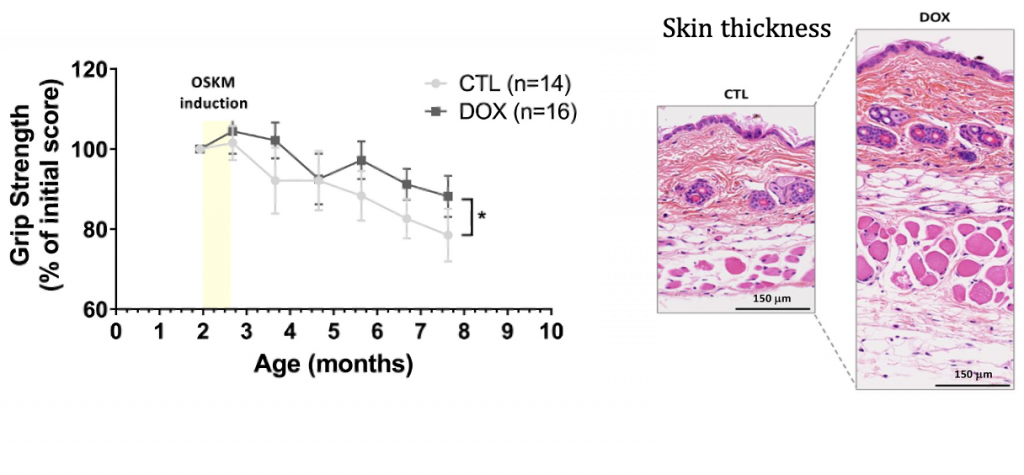
Alle and colleagues found that reprogrammed mice lose less lean mass and gain less fat mass with age compared to unprogrammed mice, indicating improvements in metabolism. They also found that reprogramming increases grip strength and skin thickness, suggesting rejuvenation of muscle and skin.
After the age accelerated mice ceased to live, Alle and collages harvested their organs to examine markers of disease. They found that cellular reprogramming mitigated much of the organ deterioration associated with human age-related diseases. For example, bone deterioration, which is associated with arthritis and osteoporosis, was largely prevented by reprogramming.
Fibrosis —characterized by inflammation and scarring — is the primary cause of age-related deterioration of organs like the lungs, kidneys, and liver. Fibrosis of the lungs is associated with a disease called idiopathic pulmonary fibrosis, which often results in respiratory failure. Alle and colleagues showed that cellular reprogramming decreased lung fibrosis. There were also signs of reduced fibrosis in the kidney and improvements in the structure of the spleen, suggesting the age reversal of multiple organs.
The Future of Anti-Aging Therapeutics?
Cellular reprogramming has previously been shown to prolong the lifespan of mice, however, these mice were extremely age accelerated mice. By showing that reprogramming can extend the lifespan of moderately age accelerated mice, Alle and colleagues reinforce the feasibility of cellular reprogramming as an anti-aging therapy. Also supporting this, other studies have shown that reprogramming can rejuvenate organs like the skin and kidneys of naturally aged mice. However, at least one study has shown that reprogramming can lead to cancer. Thus, cellular reprogramming studies are not yet ready for humans, but may be in the future.